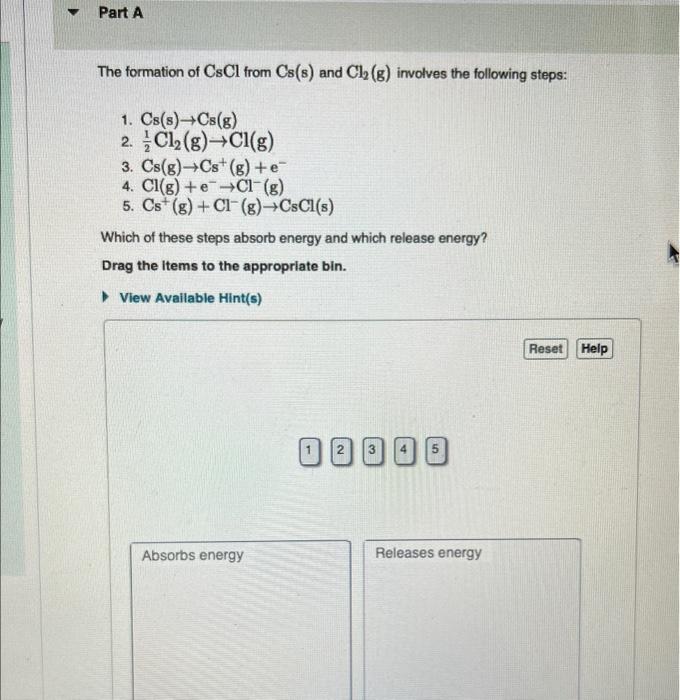
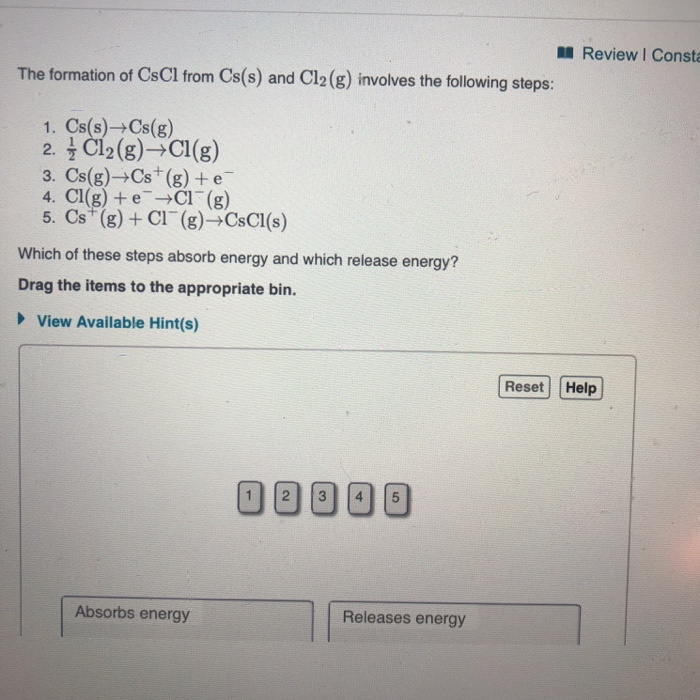
What Do Cs And Cl React To - Symbol ‘cs’ stand for the element ‘caesium ‘, ‘cs’ lies in the group one of the periodic table. What do you think is the reason for an. What do cs and cl react to form? What is a likely oxidation state of chlorine? Cs and cl react to form an ionic compound known as cesium chloride (cscl), where cs becomes a cation and cl becomes an anion held together by ionic bonds. And the halogen atom ‘chlorine’ stands for the symbol ‘cl’ lies in the group seventeenth of the periodic. How many valence electrons does na have when forming an ionic bond with cl? Cesium as the most reactive metal forms cesium halides as it reacts strongly with all. The reaction of cs with chlorine: Alkali metals are known to readily react with halogens to form ionic halides.
The reaction of cs with chlorine: What do cs and cl react to form? Symbol ‘cs’ stand for the element ‘caesium ‘, ‘cs’ lies in the group one of the periodic table. Cesium as the most reactive metal forms cesium halides as it reacts strongly with all. Cs and cl react to form an ionic compound known as cesium chloride (cscl), where cs becomes a cation and cl becomes an anion held together by ionic bonds. What is a likely oxidation state of chlorine? Alkali metals are known to readily react with halogens to form ionic halides. And the halogen atom ‘chlorine’ stands for the symbol ‘cl’ lies in the group seventeenth of the periodic. How many valence electrons does na have when forming an ionic bond with cl? What do you think is the reason for an.
Symbol ‘cs’ stand for the element ‘caesium ‘, ‘cs’ lies in the group one of the periodic table. What is a likely oxidation state of chlorine? Cesium as the most reactive metal forms cesium halides as it reacts strongly with all. What do cs and cl react to form? Alkali metals are known to readily react with halogens to form ionic halides. What do you think is the reason for an. The reaction of cs with chlorine: Cs and cl react to form an ionic compound known as cesium chloride (cscl), where cs becomes a cation and cl becomes an anion held together by ionic bonds. And the halogen atom ‘chlorine’ stands for the symbol ‘cl’ lies in the group seventeenth of the periodic. How many valence electrons does na have when forming an ionic bond with cl?
Solved The formation of CsCl from Cs(s) and Cl2( g) involves
How many valence electrons does na have when forming an ionic bond with cl? What is a likely oxidation state of chlorine? What do you think is the reason for an. The reaction of cs with chlorine: What do cs and cl react to form?
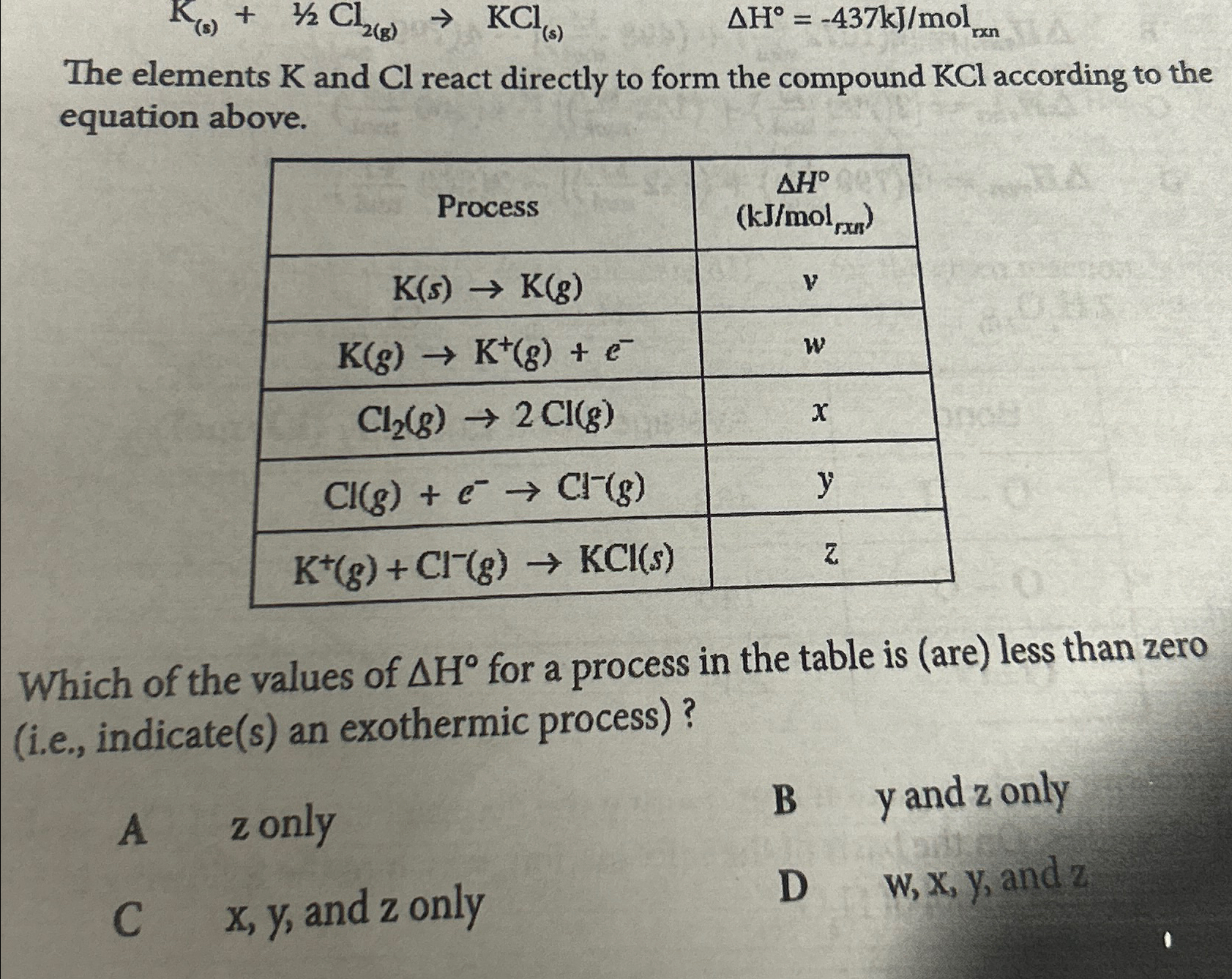
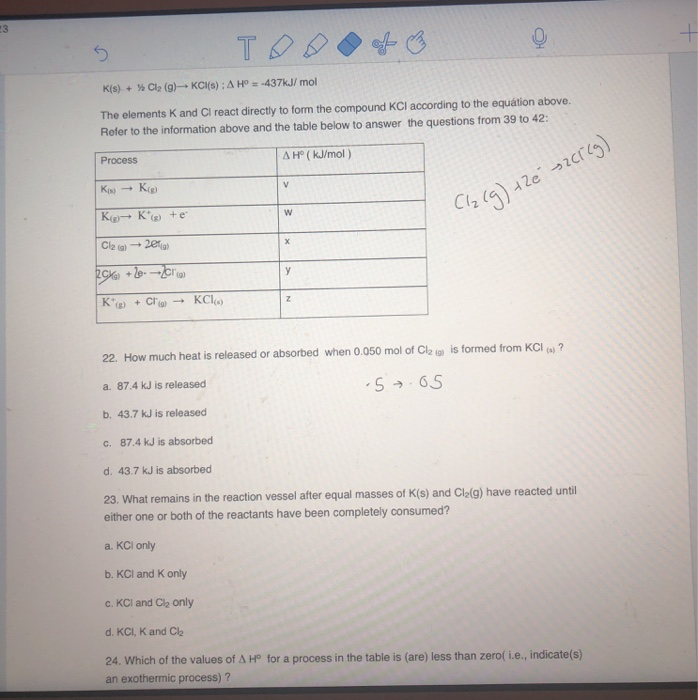
Solved The elements K and Cl react directly to form the
The reaction of cs with chlorine: And the halogen atom ‘chlorine’ stands for the symbol ‘cl’ lies in the group seventeenth of the periodic. What do you think is the reason for an. Cesium as the most reactive metal forms cesium halides as it reacts strongly with all. How many valence electrons does na have when forming an ionic bond.
CL REACT YouTube
Cs and cl react to form an ionic compound known as cesium chloride (cscl), where cs becomes a cation and cl becomes an anion held together by ionic bonds. What do cs and cl react to form? How many valence electrons does na have when forming an ionic bond with cl? What do you think is the reason for an..
Solved Review I Consta The formation of CsCl from Cs(s) and
What do you think is the reason for an. How many valence electrons does na have when forming an ionic bond with cl? Alkali metals are known to readily react with halogens to form ionic halides. Cs and cl react to form an ionic compound known as cesium chloride (cscl), where cs becomes a cation and cl becomes an anion.
CL Biography Facts, Childhood, Family Life & Achievements
The reaction of cs with chlorine: Cesium as the most reactive metal forms cesium halides as it reacts strongly with all. And the halogen atom ‘chlorine’ stands for the symbol ‘cl’ lies in the group seventeenth of the periodic. What do cs and cl react to form? Cs and cl react to form an ionic compound known as cesium chloride.
Solved K(s) + Cl (g) KCl(s) A Hº = 437kJ/mol The
What do cs and cl react to form? What do you think is the reason for an. Symbol ‘cs’ stand for the element ‘caesium ‘, ‘cs’ lies in the group one of the periodic table. What is a likely oxidation state of chlorine? Cesium as the most reactive metal forms cesium halides as it reacts strongly with all.
React Js Olajirebashitsayo Medium
And the halogen atom ‘chlorine’ stands for the symbol ‘cl’ lies in the group seventeenth of the periodic. The reaction of cs with chlorine: What is a likely oxidation state of chlorine? How many valence electrons does na have when forming an ionic bond with cl? Cs and cl react to form an ionic compound known as cesium chloride (cscl),.
Cs and Cl NMR spectroscopy and molecular dynamics modeling of
What do cs and cl react to form? What is a likely oxidation state of chlorine? Symbol ‘cs’ stand for the element ‘caesium ‘, ‘cs’ lies in the group one of the periodic table. How many valence electrons does na have when forming an ionic bond with cl? The reaction of cs with chlorine:
CL Biography Facts, Childhood, Family Life & Achievements
The reaction of cs with chlorine: How many valence electrons does na have when forming an ionic bond with cl? What is a likely oxidation state of chlorine? Cs and cl react to form an ionic compound known as cesium chloride (cscl), where cs becomes a cation and cl becomes an anion held together by ionic bonds. Symbol ‘cs’ stand.
In CsCl type structure, the coordination number of Cs+ and Cl
What do cs and cl react to form? The reaction of cs with chlorine: Cs and cl react to form an ionic compound known as cesium chloride (cscl), where cs becomes a cation and cl becomes an anion held together by ionic bonds. What do you think is the reason for an. Cesium as the most reactive metal forms cesium.
What Do You Think Is The Reason For An.
Cs and cl react to form an ionic compound known as cesium chloride (cscl), where cs becomes a cation and cl becomes an anion held together by ionic bonds. How many valence electrons does na have when forming an ionic bond with cl? Alkali metals are known to readily react with halogens to form ionic halides. And the halogen atom ‘chlorine’ stands for the symbol ‘cl’ lies in the group seventeenth of the periodic.
What Is A Likely Oxidation State Of Chlorine?
The reaction of cs with chlorine: What do cs and cl react to form? Symbol ‘cs’ stand for the element ‘caesium ‘, ‘cs’ lies in the group one of the periodic table. Cesium as the most reactive metal forms cesium halides as it reacts strongly with all.