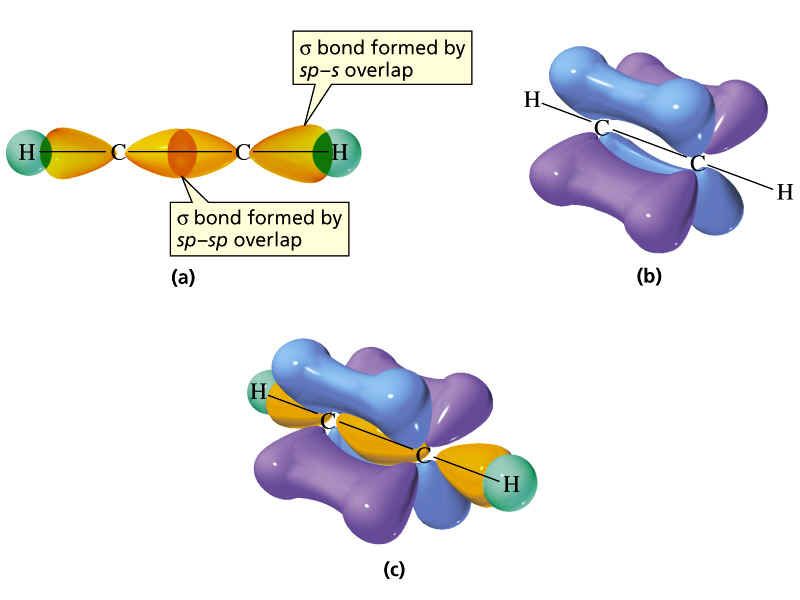
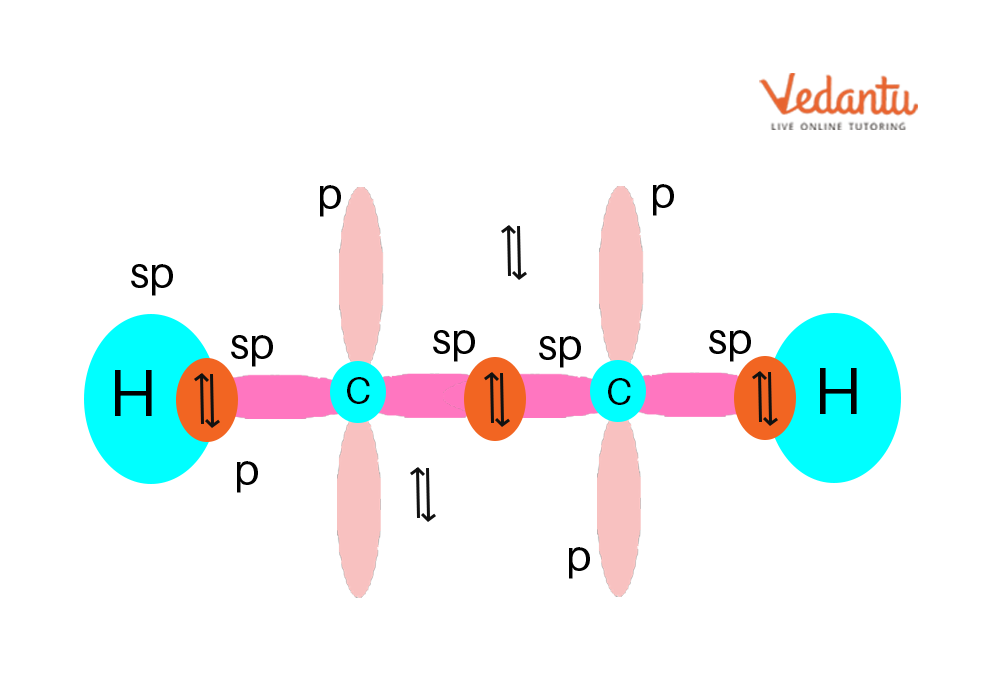
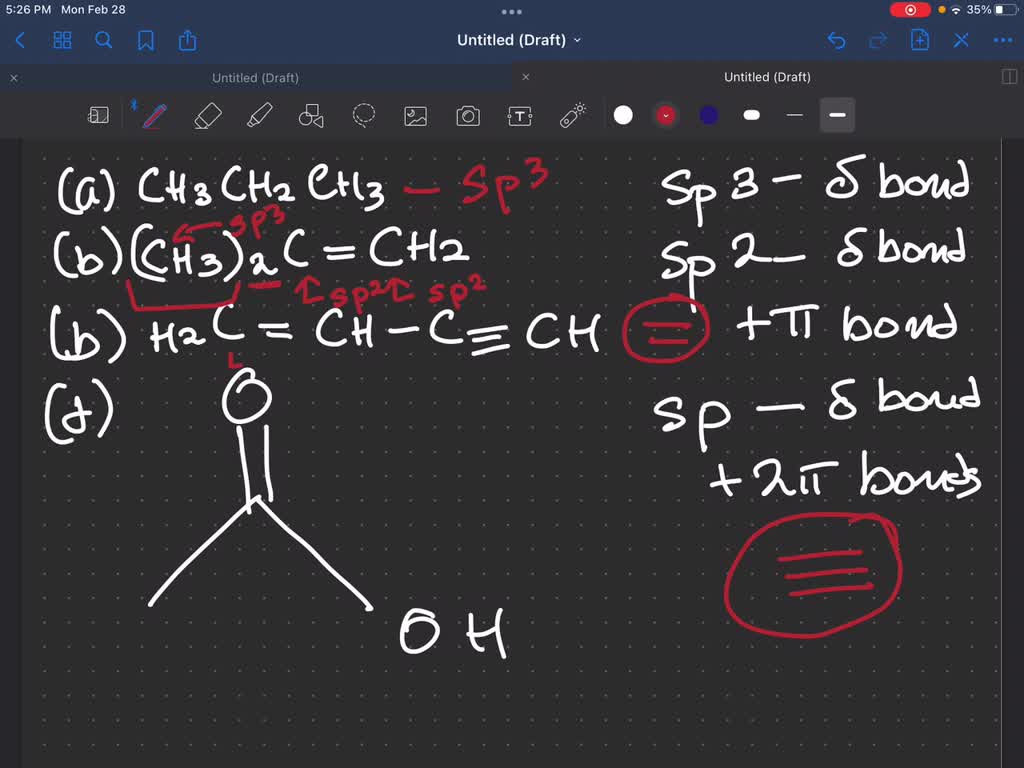
What Hybridization Would You Expect For C In Ethyne C2H2 - The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization. In ethyne (c₂h₂), each carbon atom forms two.
The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. In ethyne (c₂h₂), each carbon atom forms two. The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization.
In ethyne (c₂h₂), each carbon atom forms two. The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization.
Orbital Hybridization Chemistry Skills
The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization. The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. In ethyne (c₂h₂), each carbon atom forms two.
SOLVED Draw the lineangle structures for ethane, ethene, and ethyne
The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization. The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. In ethyne (c₂h₂), each carbon atom forms two.
32 ETHANE STRUCTURE OF ETHYNE StructureofEthane2
The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. In ethyne (c₂h₂), each carbon atom forms two. The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization.
Preparation of Ethyne Learn Important Terms and Concepts
The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization. In ethyne (c₂h₂), each carbon atom forms two.
Solved Ethyne C2H2 a. Draw the Lewis Structure of ethyne
The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization. In ethyne (c₂h₂), each carbon atom forms two.
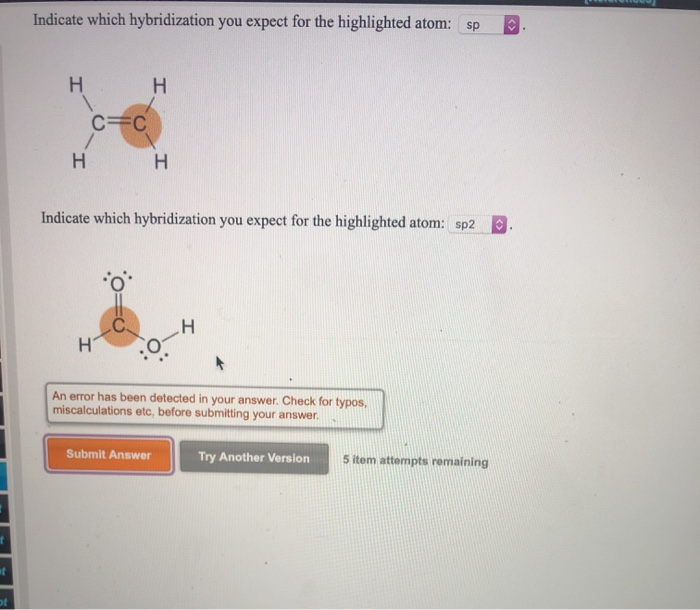
Solved Indicate which hybridization you expect for the
In ethyne (c₂h₂), each carbon atom forms two. The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization.
⏩SOLVEDWhat kind of hybridization do you expect for each carbon
The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. In ethyne (c₂h₂), each carbon atom forms two. The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization.
SOLVED Which central atom hybridization would you expect in the series
The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization. In ethyne (c₂h₂), each carbon atom forms two. The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180.
Which type of hybridization is found in the ethyne molecule?
The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. In ethyne (c₂h₂), each carbon atom forms two. The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization.
Sphybridization, definition, explanation, examples and significance
In ethyne (c₂h₂), each carbon atom forms two. The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization.
In Ethyne (C₂H₂), Each Carbon Atom Forms Two.
The sp hybridization in ethyne allows the carbon atoms to form a linear molecular geometry with a bond angle of 180. The hybridization expect for carbon in ethyne (c₂h₂) is sp atomic orbital hybridization.