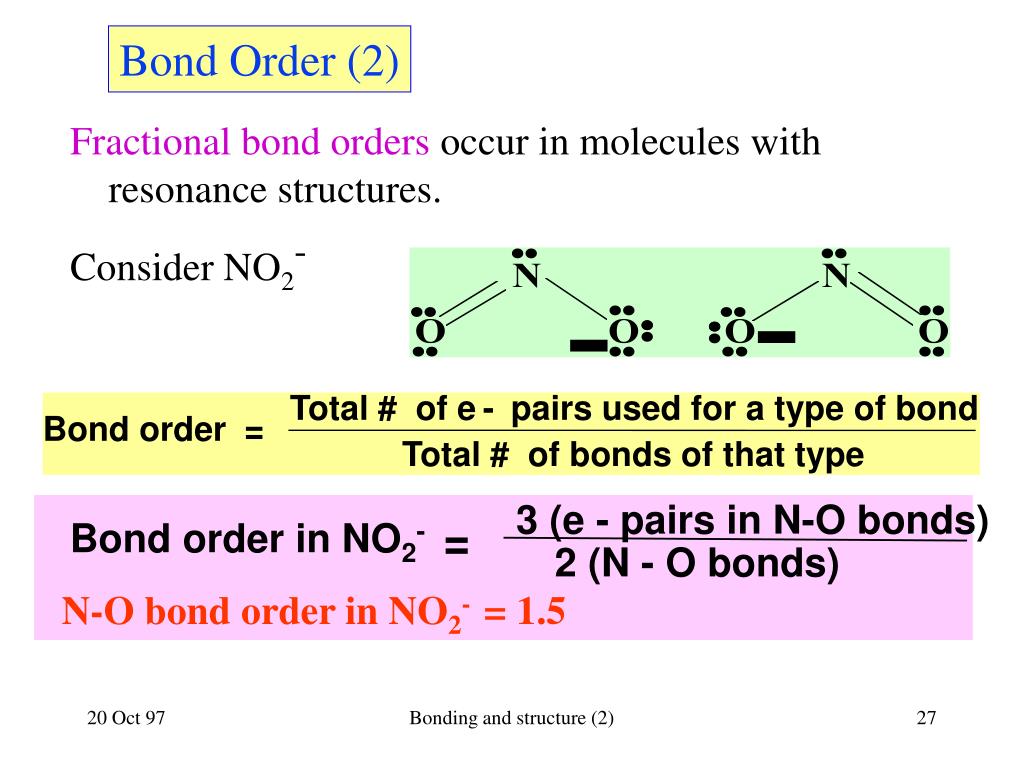
What Is The Bond Order For H2 - The bond order of h₂ is 1, indicating a stable dihydrogen molecule. Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and. For he²⁺, a hypothetical molecule, the bond order is.
Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and. For he²⁺, a hypothetical molecule, the bond order is. The bond order of h₂ is 1, indicating a stable dihydrogen molecule.
For he²⁺, a hypothetical molecule, the bond order is. The bond order of h₂ is 1, indicating a stable dihydrogen molecule. Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and.
Molecular Orbital Diagram For H2 And Bond Order
Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and. For he²⁺, a hypothetical molecule, the bond order is. The bond order of h₂ is 1, indicating a stable dihydrogen molecule.
38 The bond order of H2+ is Filo
For he²⁺, a hypothetical molecule, the bond order is. The bond order of h₂ is 1, indicating a stable dihydrogen molecule. Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and.
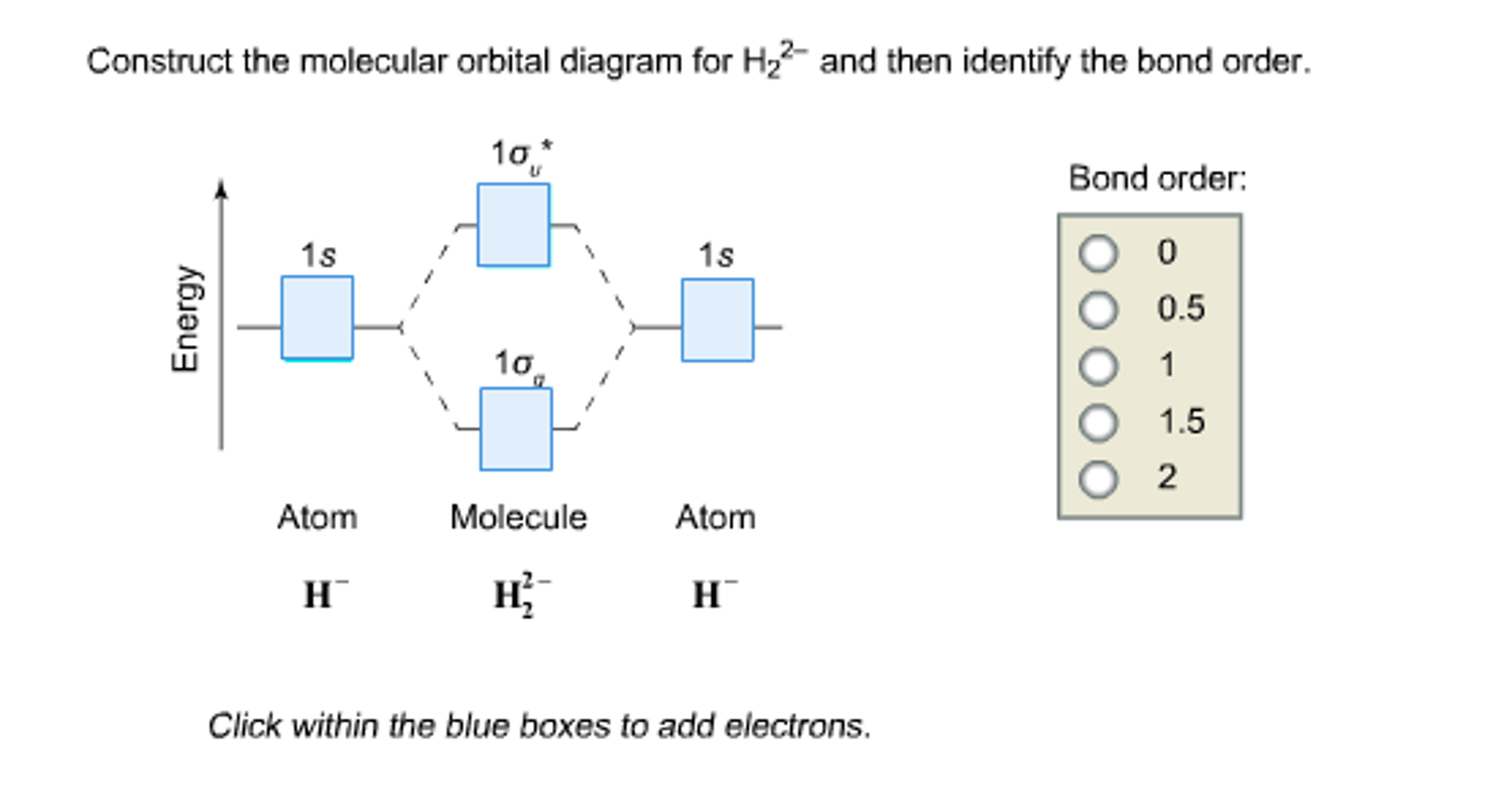
The calculated bond order in H2− ion is Filo
For he²⁺, a hypothetical molecule, the bond order is. The bond order of h₂ is 1, indicating a stable dihydrogen molecule. Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and.
Bond Order Diagram
The bond order of h₂ is 1, indicating a stable dihydrogen molecule. For he²⁺, a hypothetical molecule, the bond order is. Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and.
[Solved] what is the bond order of the hydrogen to hydrogen bond after
For he²⁺, a hypothetical molecule, the bond order is. The bond order of h₂ is 1, indicating a stable dihydrogen molecule. Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and.
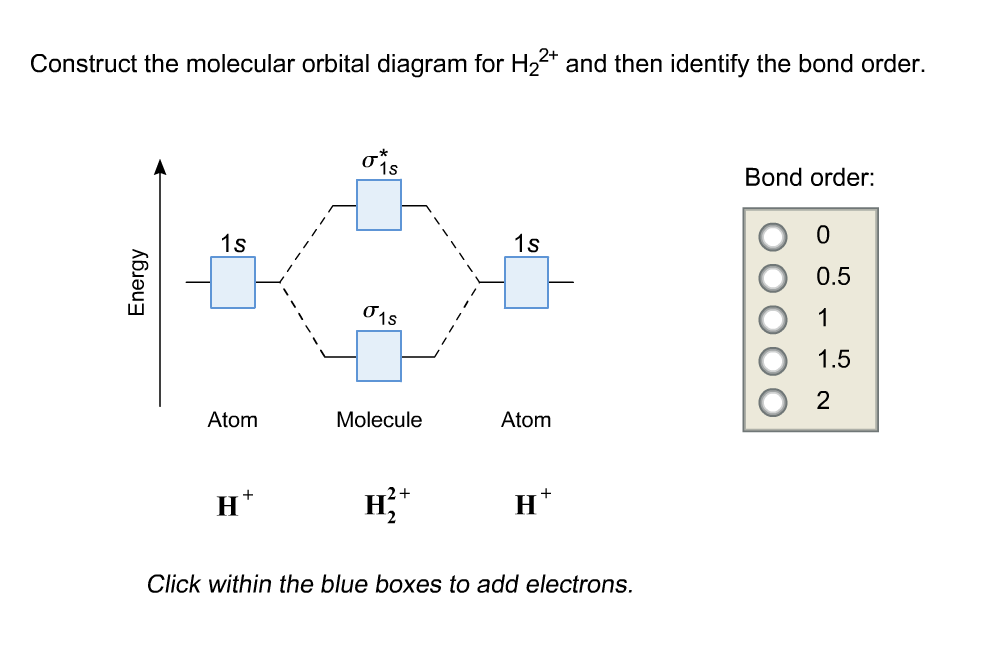
Solved Construct the molecular orbital diagram for H22+ and
For he²⁺, a hypothetical molecule, the bond order is. The bond order of h₂ is 1, indicating a stable dihydrogen molecule. Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and.
How To Calculate Bond Order From Mo Diagram General Wiring Diagram
For he²⁺, a hypothetical molecule, the bond order is. Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and. The bond order of h₂ is 1, indicating a stable dihydrogen molecule.
Define Bond Order. Calculate The Bond Order Of C2, H2 And N2. Chemistry
The bond order of h₂ is 1, indicating a stable dihydrogen molecule. Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and. For he²⁺, a hypothetical molecule, the bond order is.
What is Bond order of H2? UO Chemists
Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and. The bond order of h₂ is 1, indicating a stable dihydrogen molecule. For he²⁺, a hypothetical molecule, the bond order is.
Solved Construct the molecular orbital diagram for H_2^2
For he²⁺, a hypothetical molecule, the bond order is. The bond order of h₂ is 1, indicating a stable dihydrogen molecule. Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and.
The Bond Order Of H₂ Is 1, Indicating A Stable Dihydrogen Molecule.
For he²⁺, a hypothetical molecule, the bond order is. Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to mo theory to form one σ1s and.