What Is The Density Of Carbon Dioxide Gas At Stp - To find the density of carbon dioxide (co2) gas at standard temperature and pressure (stp), follow these steps: The density of carbon dioxide at stp is 1.964 grams per liter (g/l). Stp conditions are defined as a temperature of 0. Let's find out the density of carbon dioxide (co₂) at standard temperature and pressure (stp). What is the density of carbon dioxide at stp? 65 rows densities, molecular weight and chemical formulas of some common gases can be found in the table below: Density = mass / volume 1 mole of any ideal gas occupies a volume of 22.4 l at stp molar mass co2 = 44.01 g/mol thus density. To calculate the density of carbon dioxide at stp,. The density of carbon dioxide at stp is approximately 1.95 g/l.
Stp conditions are defined as a temperature of 0. To find the density of carbon dioxide (co2) gas at standard temperature and pressure (stp), follow these steps: What is the density of carbon dioxide at stp? 65 rows densities, molecular weight and chemical formulas of some common gases can be found in the table below: The density of carbon dioxide at stp is 1.964 grams per liter (g/l). Density = mass / volume 1 mole of any ideal gas occupies a volume of 22.4 l at stp molar mass co2 = 44.01 g/mol thus density. To calculate the density of carbon dioxide at stp,. The density of carbon dioxide at stp is approximately 1.95 g/l. Let's find out the density of carbon dioxide (co₂) at standard temperature and pressure (stp).
Let's find out the density of carbon dioxide (co₂) at standard temperature and pressure (stp). Density = mass / volume 1 mole of any ideal gas occupies a volume of 22.4 l at stp molar mass co2 = 44.01 g/mol thus density. The density of carbon dioxide at stp is 1.964 grams per liter (g/l). To calculate the density of carbon dioxide at stp,. 65 rows densities, molecular weight and chemical formulas of some common gases can be found in the table below: To find the density of carbon dioxide (co2) gas at standard temperature and pressure (stp), follow these steps: What is the density of carbon dioxide at stp? Stp conditions are defined as a temperature of 0. The density of carbon dioxide at stp is approximately 1.95 g/l.
27. The density of a gas STP is 1.5g/L STP. Its molecular weight is 1
Stp conditions are defined as a temperature of 0. Density = mass / volume 1 mole of any ideal gas occupies a volume of 22.4 l at stp molar mass co2 = 44.01 g/mol thus density. The density of carbon dioxide at stp is approximately 1.95 g/l. To calculate the density of carbon dioxide at stp,. The density of carbon.
What Is the Density of Carbon Dioxide at Stp
65 rows densities, molecular weight and chemical formulas of some common gases can be found in the table below: What is the density of carbon dioxide at stp? The density of carbon dioxide at stp is approximately 1.95 g/l. To find the density of carbon dioxide (co2) gas at standard temperature and pressure (stp), follow these steps: Density = mass.
SOLVED Assuming it behaves as an ideal gas, calculate the density of
65 rows densities, molecular weight and chemical formulas of some common gases can be found in the table below: To find the density of carbon dioxide (co2) gas at standard temperature and pressure (stp), follow these steps: Let's find out the density of carbon dioxide (co₂) at standard temperature and pressure (stp). Stp conditions are defined as a temperature of.
SOLVED Calculate the density of carbon dioxide at STP Enter your
The density of carbon dioxide at stp is approximately 1.95 g/l. To find the density of carbon dioxide (co2) gas at standard temperature and pressure (stp), follow these steps: Density = mass / volume 1 mole of any ideal gas occupies a volume of 22.4 l at stp molar mass co2 = 44.01 g/mol thus density. What is the density.
Carbon Dioxide Density, Heat Capacity, Thermal Conductivity
Let's find out the density of carbon dioxide (co₂) at standard temperature and pressure (stp). Density = mass / volume 1 mole of any ideal gas occupies a volume of 22.4 l at stp molar mass co2 = 44.01 g/mol thus density. The density of carbon dioxide at stp is approximately 1.95 g/l. What is the density of carbon dioxide.
Solved What is the density of carbon monoxide gas at STP?
To find the density of carbon dioxide (co2) gas at standard temperature and pressure (stp), follow these steps: To calculate the density of carbon dioxide at stp,. 65 rows densities, molecular weight and chemical formulas of some common gases can be found in the table below: The density of carbon dioxide at stp is approximately 1.95 g/l. Stp conditions are.
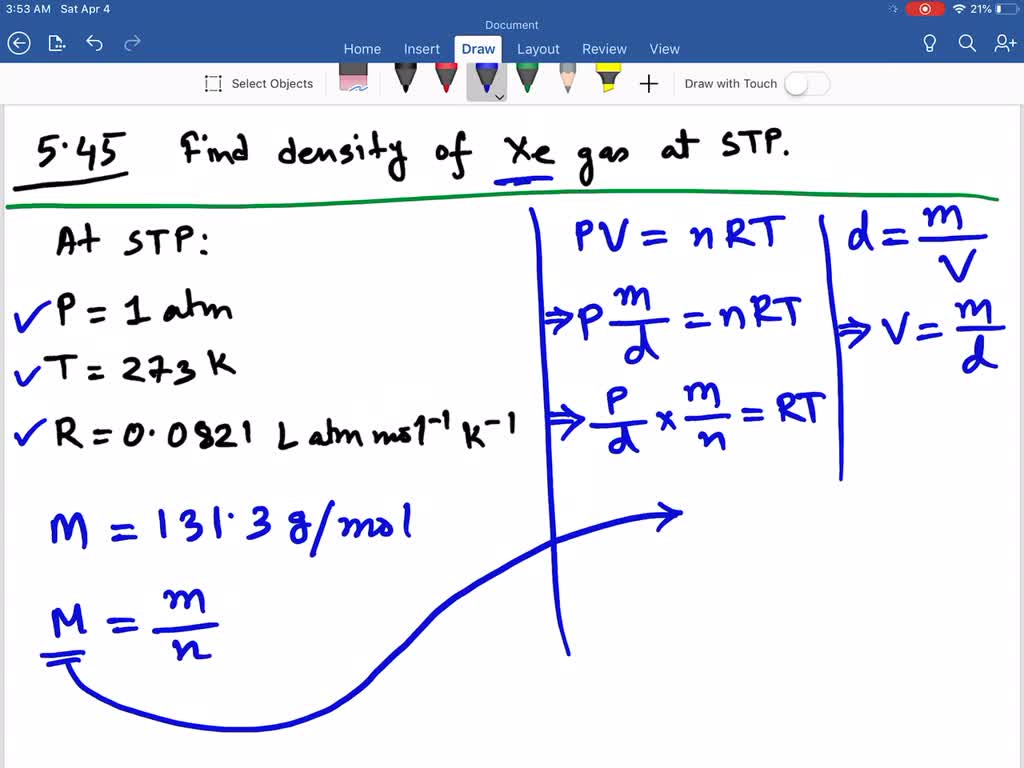
SOLVEDWhat is the density of Xe gas at STP?
The density of carbon dioxide at stp is 1.964 grams per liter (g/l). Stp conditions are defined as a temperature of 0. 65 rows densities, molecular weight and chemical formulas of some common gases can be found in the table below: The density of carbon dioxide at stp is approximately 1.95 g/l. What is the density of carbon dioxide at.
Density of Carbon Dioxide khawkins17
To calculate the density of carbon dioxide at stp,. What is the density of carbon dioxide at stp? The density of carbon dioxide at stp is 1.964 grams per liter (g/l). Let's find out the density of carbon dioxide (co₂) at standard temperature and pressure (stp). The density of carbon dioxide at stp is approximately 1.95 g/l.
SOLVED How much carbon dioxide gas, in grams, is in a 1.0L balloon at
The density of carbon dioxide at stp is 1.964 grams per liter (g/l). Let's find out the density of carbon dioxide (co₂) at standard temperature and pressure (stp). To calculate the density of carbon dioxide at stp,. Density = mass / volume 1 mole of any ideal gas occupies a volume of 22.4 l at stp molar mass co2 =.
SOLVED Use the molar volume of a gas at STP to calculate the density
To calculate the density of carbon dioxide at stp,. Stp conditions are defined as a temperature of 0. What is the density of carbon dioxide at stp? 65 rows densities, molecular weight and chemical formulas of some common gases can be found in the table below: Density = mass / volume 1 mole of any ideal gas occupies a volume.
What Is The Density Of Carbon Dioxide At Stp?
The density of carbon dioxide at stp is approximately 1.95 g/l. 65 rows densities, molecular weight and chemical formulas of some common gases can be found in the table below: To find the density of carbon dioxide (co2) gas at standard temperature and pressure (stp), follow these steps: The density of carbon dioxide at stp is 1.964 grams per liter (g/l).
To Calculate The Density Of Carbon Dioxide At Stp,.
Stp conditions are defined as a temperature of 0. Let's find out the density of carbon dioxide (co₂) at standard temperature and pressure (stp). Density = mass / volume 1 mole of any ideal gas occupies a volume of 22.4 l at stp molar mass co2 = 44.01 g/mol thus density.