What Is The Oxidation Number Of Hg In Hg2Cl2 - This can be determined by considering the oxidation states of other. (1) assume the element to be x and since it is hg2 take it 2x. In the case of hg₂cl₂, the oxidation state of mercury is +1. The oxidation number of hg in hg2cl2 is +1. Lets say hg2 for reactants (+1. You will look at the oxidation numbers of the reactant and see how it changed in the product. To calculate the oxidation state of hg in hg2cl2.
In the case of hg₂cl₂, the oxidation state of mercury is +1. The oxidation number of hg in hg2cl2 is +1. (1) assume the element to be x and since it is hg2 take it 2x. This can be determined by considering the oxidation states of other. Lets say hg2 for reactants (+1. To calculate the oxidation state of hg in hg2cl2. You will look at the oxidation numbers of the reactant and see how it changed in the product.
(1) assume the element to be x and since it is hg2 take it 2x. You will look at the oxidation numbers of the reactant and see how it changed in the product. This can be determined by considering the oxidation states of other. Lets say hg2 for reactants (+1. The oxidation number of hg in hg2cl2 is +1. In the case of hg₂cl₂, the oxidation state of mercury is +1. To calculate the oxidation state of hg in hg2cl2.
SOLVED What is the oxidation number of Hg in Hg2Cl2? a) 2 b) 0 c) 1
Lets say hg2 for reactants (+1. (1) assume the element to be x and since it is hg2 take it 2x. The oxidation number of hg in hg2cl2 is +1. You will look at the oxidation numbers of the reactant and see how it changed in the product. In the case of hg₂cl₂, the oxidation state of mercury is +1.
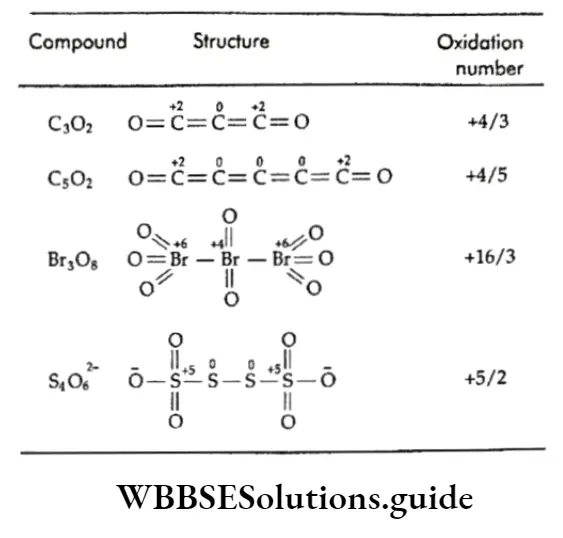
Oxidation Number Definition, Calculation and Examples WBBSE Solutions
To calculate the oxidation state of hg in hg2cl2. (1) assume the element to be x and since it is hg2 take it 2x. Lets say hg2 for reactants (+1. In the case of hg₂cl₂, the oxidation state of mercury is +1. This can be determined by considering the oxidation states of other.
Oxidation number of chlorine atoms in CaOCl2 are Filo
The oxidation number of hg in hg2cl2 is +1. You will look at the oxidation numbers of the reactant and see how it changed in the product. This can be determined by considering the oxidation states of other. To calculate the oxidation state of hg in hg2cl2. (1) assume the element to be x and since it is hg2 take.
How To Balance A Chemical Equation By Oxidation Number Method In Tamil
The oxidation number of hg in hg2cl2 is +1. You will look at the oxidation numbers of the reactant and see how it changed in the product. (1) assume the element to be x and since it is hg2 take it 2x. To calculate the oxidation state of hg in hg2cl2. This can be determined by considering the oxidation states.
OXIDATION NUMBER PPT PPT Free Download
Lets say hg2 for reactants (+1. (1) assume the element to be x and since it is hg2 take it 2x. You will look at the oxidation numbers of the reactant and see how it changed in the product. In the case of hg₂cl₂, the oxidation state of mercury is +1. To calculate the oxidation state of hg in hg2cl2.
Rate of Hg 0 photocatalytic oxidation vs. inlet Hg 0 concentration
(1) assume the element to be x and since it is hg2 take it 2x. In the case of hg₂cl₂, the oxidation state of mercury is +1. The oxidation number of hg in hg2cl2 is +1. This can be determined by considering the oxidation states of other. You will look at the oxidation numbers of the reactant and see how.
Hg oxidation with Cl 2 in N 2 and CO 2 . Download Scientific Diagram
Lets say hg2 for reactants (+1. The oxidation number of hg in hg2cl2 is +1. In the case of hg₂cl₂, the oxidation state of mercury is +1. You will look at the oxidation numbers of the reactant and see how it changed in the product. (1) assume the element to be x and since it is hg2 take it 2x.
Plot of Observed Hg Oxidation Versus Predicted Hg Oxidation, Using
This can be determined by considering the oxidation states of other. Lets say hg2 for reactants (+1. The oxidation number of hg in hg2cl2 is +1. In the case of hg₂cl₂, the oxidation state of mercury is +1. (1) assume the element to be x and since it is hg2 take it 2x.
Oxidation Number Rules Chart
You will look at the oxidation numbers of the reactant and see how it changed in the product. (1) assume the element to be x and since it is hg2 take it 2x. Lets say hg2 for reactants (+1. This can be determined by considering the oxidation states of other. To calculate the oxidation state of hg in hg2cl2.
Oxidation number of 'Co' in Hg[Co(SCN)4]
To calculate the oxidation state of hg in hg2cl2. The oxidation number of hg in hg2cl2 is +1. This can be determined by considering the oxidation states of other. In the case of hg₂cl₂, the oxidation state of mercury is +1. You will look at the oxidation numbers of the reactant and see how it changed in the product.
The Oxidation Number Of Hg In Hg2Cl2 Is +1.
In the case of hg₂cl₂, the oxidation state of mercury is +1. You will look at the oxidation numbers of the reactant and see how it changed in the product. (1) assume the element to be x and since it is hg2 take it 2x. To calculate the oxidation state of hg in hg2cl2.
This Can Be Determined By Considering The Oxidation States Of Other.
Lets say hg2 for reactants (+1.








![Oxidation number of 'Co' in Hg[Co(SCN)4]](https://d1hj4to4g9ba46.cloudfront.net/questions/2021628_1318215_ans_11fe87bacafb4aefbff9e7364e9f75f0.jpg)