What Is The Oxidation State Of Carbon In Co32 - (a) 1+ (b) 2+ (c). Therefore, the oxidation state of $$c, is \ iv$$. The sum of the formal oxidation numbers, in a complex or compound ion, must. The sum of the formal oxidation numbers, in a. The carbon is fully oxidized. Therefore, the oxidation state of c, i s i v. The oxidation number of carbon in the carbonate ion (co₃²⁻) can be determined by knowing the oxidation number of oxygen and. The oxidation number of carbon in co32− can be calculated by knowing the oxidation numbers of the atoms it's bonded to and the. What is the formal charge of carbon on co? This is because the sum of the oxidation numbers in the carbonate ion must.
(a) 1+ (b) 2+ (c). The sum of the formal oxidation numbers, in a. The oxidation number of carbon in co32− can be calculated by knowing the oxidation numbers of the atoms it's bonded to and the. The sum of the formal oxidation numbers, in a complex or compound ion, must. Therefore, the oxidation state of $$c, is \ iv$$. The oxidation number of carbon in the carbonate ion (co₃²⁻) can be determined by knowing the oxidation number of oxygen and. What is the formal charge of carbon on co? The carbon is fully oxidized. Therefore, the oxidation state of c, i s i v. This is because the sum of the oxidation numbers in the carbonate ion must.
The sum of the formal oxidation numbers, in a. The sum of the formal oxidation numbers, in a complex or compound ion, must. The oxidation number of carbon in the carbonate ion (co₃²⁻) can be determined by knowing the oxidation number of oxygen and. Therefore, the oxidation state of $$c, is \ iv$$. The carbon is fully oxidized. What is the formal charge of carbon on co? (a) 1+ (b) 2+ (c). The oxidation number of carbon in co32− can be calculated by knowing the oxidation numbers of the atoms it's bonded to and the. Therefore, the oxidation state of c, i s i v. This is because the sum of the oxidation numbers in the carbonate ion must.
Oxidation States of Carbon Wolfram Demonstrations Project
The oxidation number of carbon in the carbonate ion (co₃²⁻) can be determined by knowing the oxidation number of oxygen and. This is because the sum of the oxidation numbers in the carbonate ion must. What is the formal charge of carbon on co? (a) 1+ (b) 2+ (c). The sum of the formal oxidation numbers, in a complex or.
SOLVED 'What is the oxidation state of carbon in CO?'
The carbon is fully oxidized. The sum of the formal oxidation numbers, in a complex or compound ion, must. The oxidation number of carbon in the carbonate ion (co₃²⁻) can be determined by knowing the oxidation number of oxygen and. Therefore, the oxidation state of c, i s i v. Therefore, the oxidation state of $$c, is \ iv$$.
Periodic Table Oxidation State Carbon Chemical Element Hydrogen, PNG, 990x1401px, Periodic Table
The carbon is fully oxidized. Therefore, the oxidation state of $$c, is \ iv$$. Therefore, the oxidation state of c, i s i v. What is the formal charge of carbon on co? The sum of the formal oxidation numbers, in a complex or compound ion, must.
Oxidation State
The sum of the formal oxidation numbers, in a complex or compound ion, must. (a) 1+ (b) 2+ (c). The carbon is fully oxidized. The sum of the formal oxidation numbers, in a. The oxidation number of carbon in the carbonate ion (co₃²⁻) can be determined by knowing the oxidation number of oxygen and.
Oxidation State of Carbon in Organic Compound
This is because the sum of the oxidation numbers in the carbonate ion must. The carbon is fully oxidized. Therefore, the oxidation state of c, i s i v. (a) 1+ (b) 2+ (c). Therefore, the oxidation state of $$c, is \ iv$$.
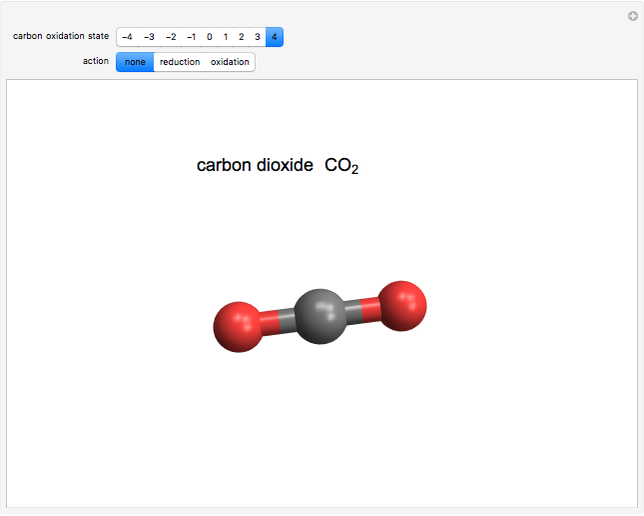
What is the oxidation state of carbon in carbondioxide?
Therefore, the oxidation state of $$c, is \ iv$$. The carbon is fully oxidized. The sum of the formal oxidation numbers, in a. This is because the sum of the oxidation numbers in the carbonate ion must. What is the formal charge of carbon on co?
Oxidation State of Carbon in Organic Compound
The oxidation number of carbon in co32− can be calculated by knowing the oxidation numbers of the atoms it's bonded to and the. The sum of the formal oxidation numbers, in a complex or compound ion, must. Therefore, the oxidation state of $$c, is \ iv$$. The oxidation number of carbon in the carbonate ion (co₃²⁻) can be determined by.
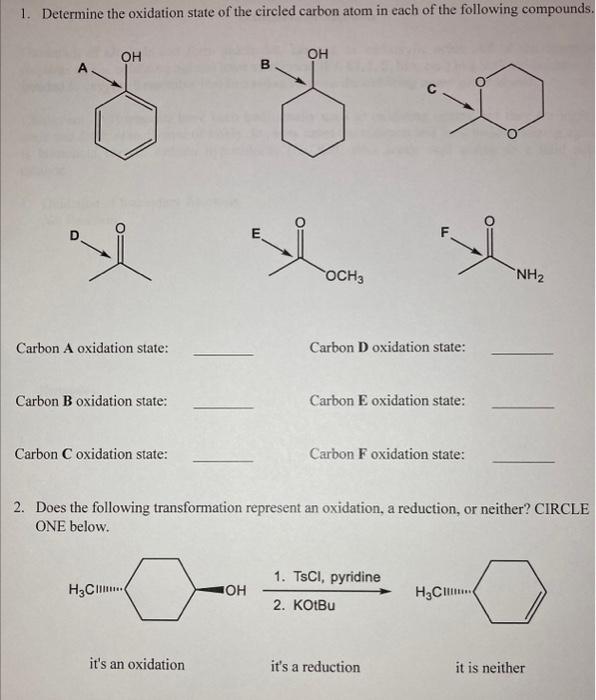
Solved 1. Determine the oxidation state of the circled
(a) 1+ (b) 2+ (c). The sum of the formal oxidation numbers, in a. This is because the sum of the oxidation numbers in the carbonate ion must. Therefore, the oxidation state of $$c, is \ iv$$. The sum of the formal oxidation numbers, in a complex or compound ion, must.
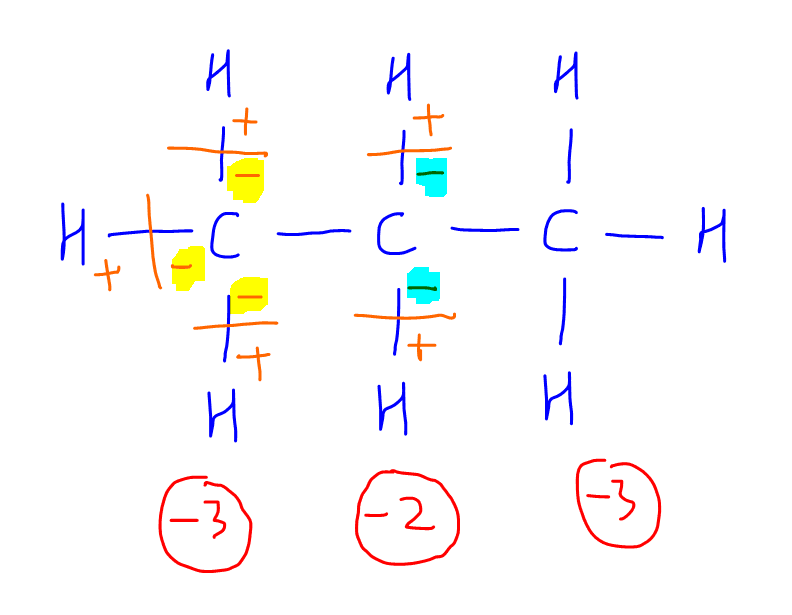
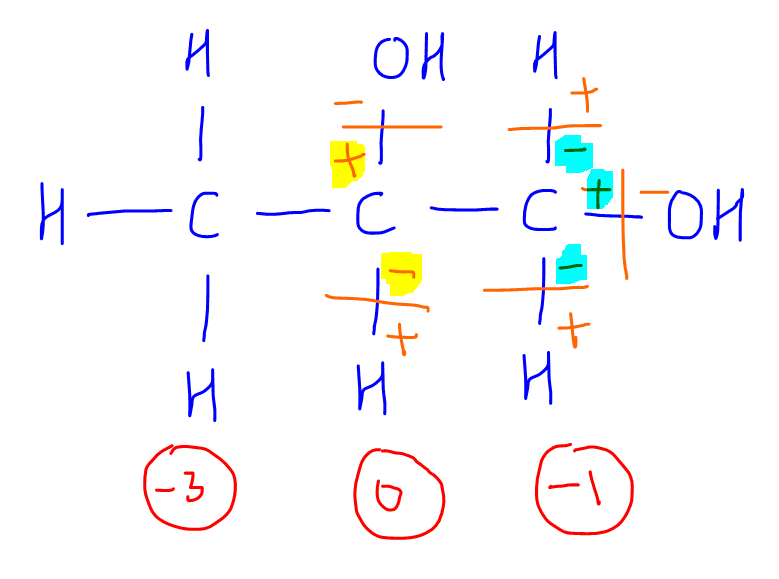
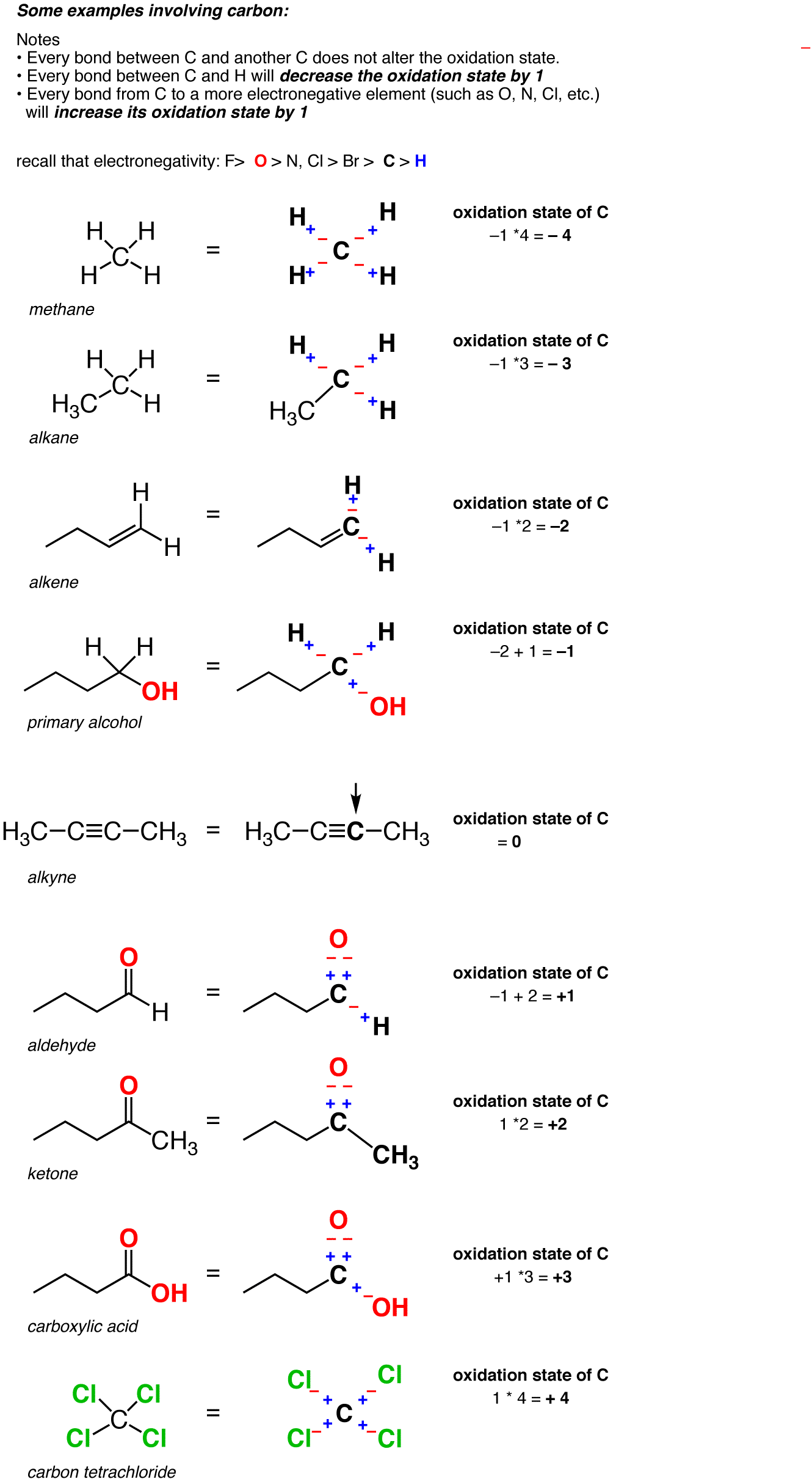
Calculating the oxidation state of a carbon Master Organic Chemistry
The carbon is fully oxidized. The oxidation number of carbon in the carbonate ion (co₃²⁻) can be determined by knowing the oxidation number of oxygen and. This is because the sum of the oxidation numbers in the carbonate ion must. Therefore, the oxidation state of c, i s i v. The sum of the formal oxidation numbers, in a complex.
Co2 Oxidation State
Therefore, the oxidation state of $$c, is \ iv$$. Therefore, the oxidation state of c, i s i v. The oxidation number of carbon in the carbonate ion (co₃²⁻) can be determined by knowing the oxidation number of oxygen and. The oxidation number of carbon in co32− can be calculated by knowing the oxidation numbers of the atoms it's bonded.
The Oxidation Number Of Carbon In The Carbonate Ion (Co₃²⁻) Can Be Determined By Knowing The Oxidation Number Of Oxygen And.
What is the formal charge of carbon on co? The carbon is fully oxidized. (a) 1+ (b) 2+ (c). Therefore, the oxidation state of $$c, is \ iv$$.
The Oxidation Number Of Carbon In Co32− Can Be Calculated By Knowing The Oxidation Numbers Of The Atoms It's Bonded To And The.
Therefore, the oxidation state of c, i s i v. The sum of the formal oxidation numbers, in a complex or compound ion, must. The sum of the formal oxidation numbers, in a. This is because the sum of the oxidation numbers in the carbonate ion must.