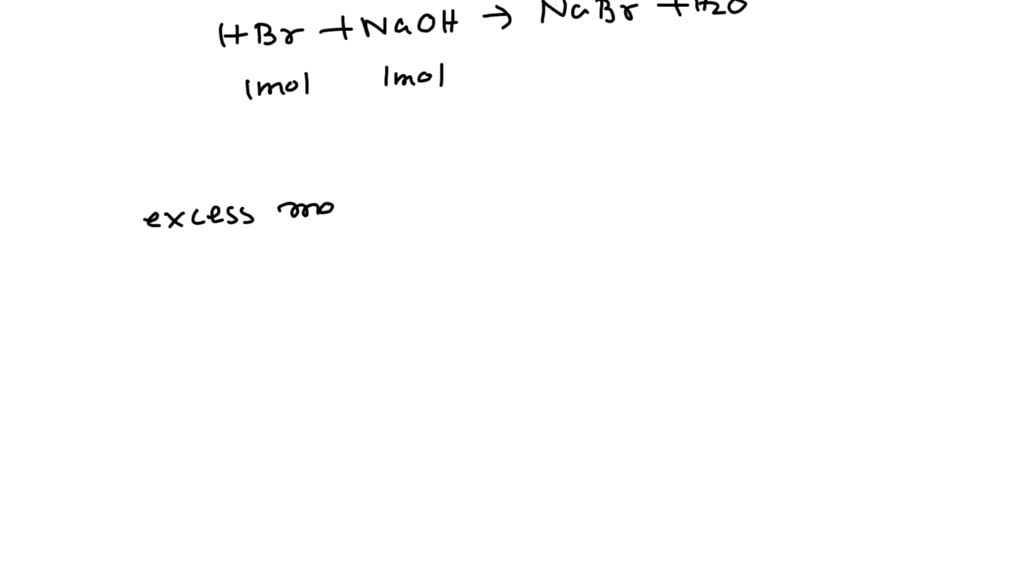What Is The Ph Of A 6 50X10 3 M Koh Solution - We'll begin by writing the balanced dissociation equation for koh. Next, we shall determine the poh of the solution. Ph is a measure of how acidic or basic a solution is. Phs with less than 7. Ph has a range from 0 to 14.
We'll begin by writing the balanced dissociation equation for koh. Ph is a measure of how acidic or basic a solution is. Phs with less than 7. Ph has a range from 0 to 14. Next, we shall determine the poh of the solution.
Phs with less than 7. Next, we shall determine the poh of the solution. Ph has a range from 0 to 14. We'll begin by writing the balanced dissociation equation for koh. Ph is a measure of how acidic or basic a solution is.
SOLVED Calculate the pH of a 2.8 x 10^3 M KOH solution.
We'll begin by writing the balanced dissociation equation for koh. Phs with less than 7. Ph is a measure of how acidic or basic a solution is. Ph has a range from 0 to 14. Next, we shall determine the poh of the solution.
SOLVED determine the pH of a 0.04 M KOH solution
Next, we shall determine the poh of the solution. We'll begin by writing the balanced dissociation equation for koh. Ph is a measure of how acidic or basic a solution is. Phs with less than 7. Ph has a range from 0 to 14.
Solved A 50.0 mL solution of 0.165 M KOH is titrated with
Phs with less than 7. Ph is a measure of how acidic or basic a solution is. Ph has a range from 0 to 14. Next, we shall determine the poh of the solution. We'll begin by writing the balanced dissociation equation for koh.
Solved 1. What does the pH of a solution tell you? 2. What
Ph has a range from 0 to 14. We'll begin by writing the balanced dissociation equation for koh. Ph is a measure of how acidic or basic a solution is. Phs with less than 7. Next, we shall determine the poh of the solution.
SOLVED Calculate the pH of 100.00 mL of 0.10 M KOH solution after 145.
Ph has a range from 0 to 14. We'll begin by writing the balanced dissociation equation for koh. Ph is a measure of how acidic or basic a solution is. Phs with less than 7. Next, we shall determine the poh of the solution.
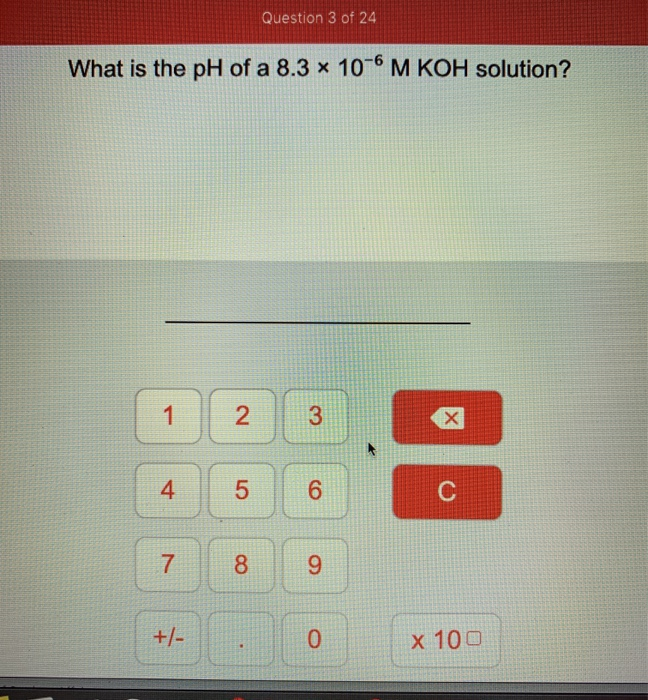
Solved what is the pH of a 8.3 x 10^6 M KOH solution
Phs with less than 7. We'll begin by writing the balanced dissociation equation for koh. Next, we shall determine the poh of the solution. Ph is a measure of how acidic or basic a solution is. Ph has a range from 0 to 14.
Solved Determine the pH in a 2.58 x 103 M KOH solution at
Phs with less than 7. Ph has a range from 0 to 14. Next, we shall determine the poh of the solution. Ph is a measure of how acidic or basic a solution is. We'll begin by writing the balanced dissociation equation for koh.
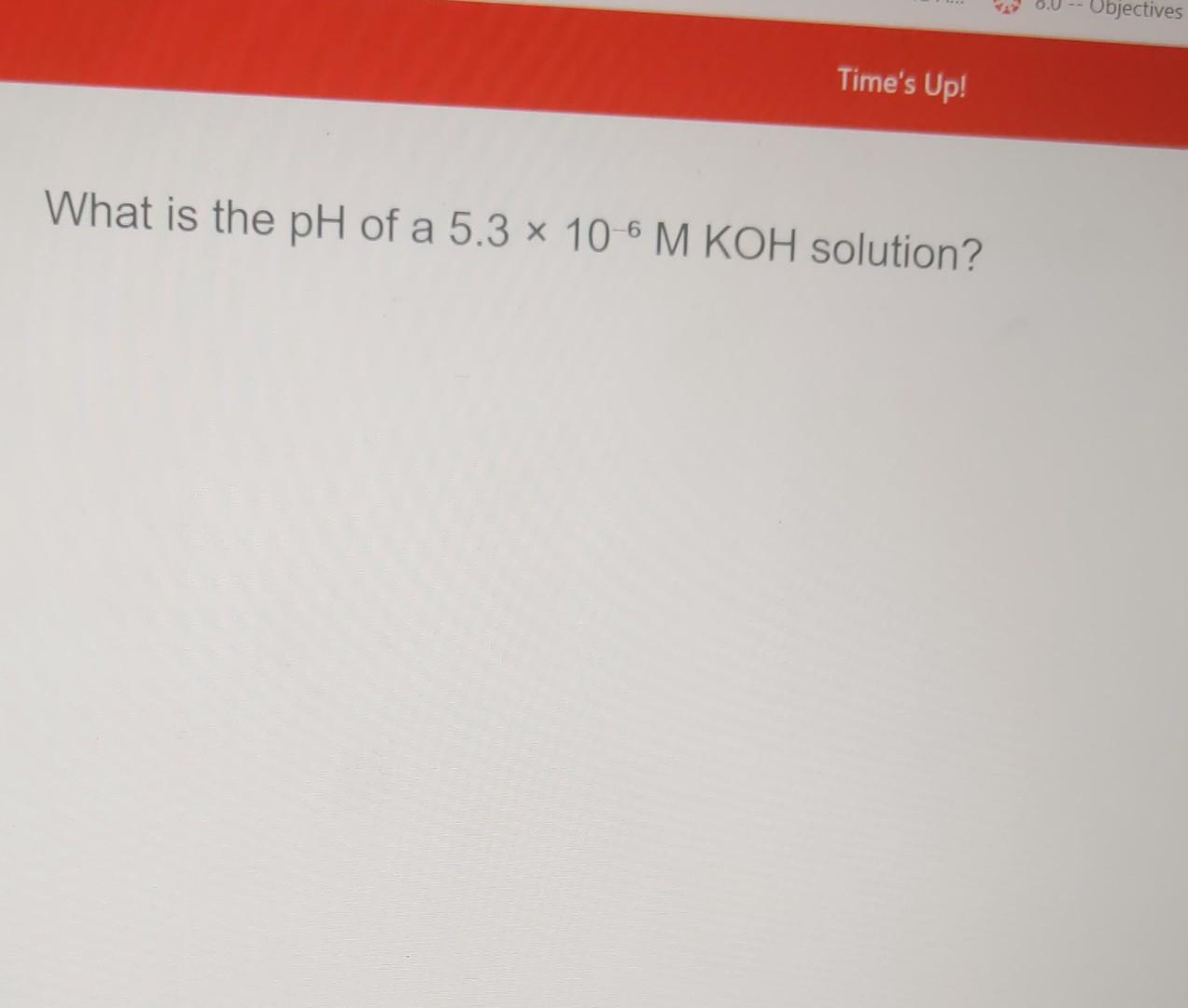
Solved What is the pH of a 5.3×10−6MKOH solution?
We'll begin by writing the balanced dissociation equation for koh. Ph is a measure of how acidic or basic a solution is. Phs with less than 7. Ph has a range from 0 to 14. Next, we shall determine the poh of the solution.
At 90 ^o C, the pH of 0.001M KOH solution will be
Phs with less than 7. We'll begin by writing the balanced dissociation equation for koh. Ph is a measure of how acidic or basic a solution is. Next, we shall determine the poh of the solution. Ph has a range from 0 to 14.
Phs With Less Than 7.
We'll begin by writing the balanced dissociation equation for koh. Ph has a range from 0 to 14. Ph is a measure of how acidic or basic a solution is. Next, we shall determine the poh of the solution.