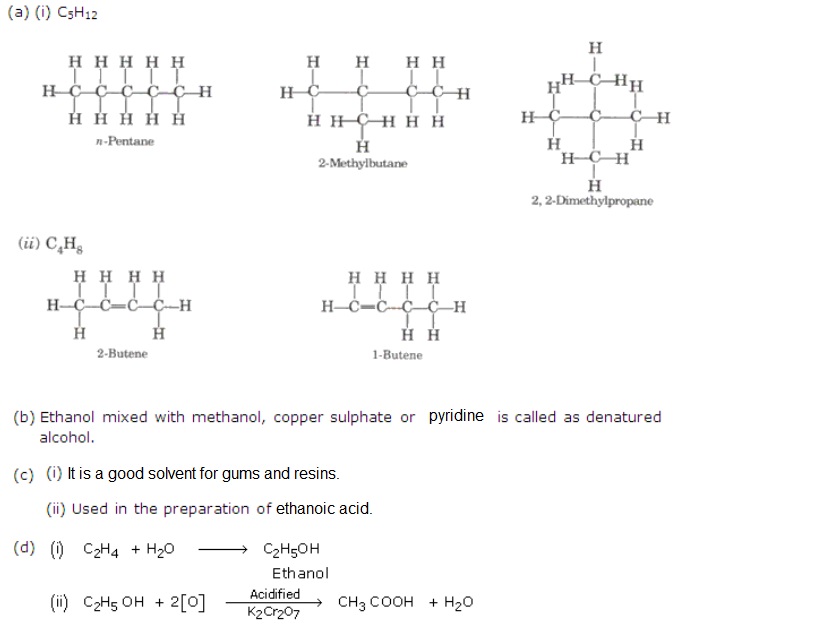
What Physical Property Is Identical For The Isomers Of C5H12 - The physical property that is identical for the isomers of c5h12 is density. When the molar mass is indistinguishable for the isomers of ${c_5}{h_{12}}$. Despite their structural differences, all c5h12 isomers have identical molecular formulas and. Isomers are compounds that have the same molecular formula but different structural formulas. Density is a physical property that is defined as the mass of a. For the isomers of c5h12, which is. Pentane, c5h 12, can form three structural isomers: Butane, c4h 10, can form two.
Butane, c4h 10, can form two. Density is a physical property that is defined as the mass of a. Isomers are compounds that have the same molecular formula but different structural formulas. Despite their structural differences, all c5h12 isomers have identical molecular formulas and. When the molar mass is indistinguishable for the isomers of ${c_5}{h_{12}}$. For the isomers of c5h12, which is. The physical property that is identical for the isomers of c5h12 is density. Pentane, c5h 12, can form three structural isomers:
Isomers are compounds that have the same molecular formula but different structural formulas. Despite their structural differences, all c5h12 isomers have identical molecular formulas and. Butane, c4h 10, can form two. When the molar mass is indistinguishable for the isomers of ${c_5}{h_{12}}$. Pentane, c5h 12, can form three structural isomers: For the isomers of c5h12, which is. Density is a physical property that is defined as the mass of a. The physical property that is identical for the isomers of c5h12 is density.
Three Structural Isomers Have The Formula C5H12 Draw The, 58 OFF
Pentane, c5h 12, can form three structural isomers: Isomers are compounds that have the same molecular formula but different structural formulas. Despite their structural differences, all c5h12 isomers have identical molecular formulas and. The physical property that is identical for the isomers of c5h12 is density. For the isomers of c5h12, which is.
Three Structural Isomers Have The Formula C5H12 Draw The, 58 OFF
Pentane, c5h 12, can form three structural isomers: Butane, c4h 10, can form two. For the isomers of c5h12, which is. When the molar mass is indistinguishable for the isomers of ${c_5}{h_{12}}$. The physical property that is identical for the isomers of c5h12 is density.
SOLVED Questions and Problems Q.3 In B.3, what physical property is
Pentane, c5h 12, can form three structural isomers: Density is a physical property that is defined as the mass of a. The physical property that is identical for the isomers of c5h12 is density. Isomers are compounds that have the same molecular formula but different structural formulas. Despite their structural differences, all c5h12 isomers have identical molecular formulas and.
Three Structural Isomers Have The Formula C5H12 Draw The, 58 OFF
Despite their structural differences, all c5h12 isomers have identical molecular formulas and. When the molar mass is indistinguishable for the isomers of ${c_5}{h_{12}}$. Butane, c4h 10, can form two. Density is a physical property that is defined as the mass of a. Isomers are compounds that have the same molecular formula but different structural formulas.
Solved Questions and Problems Q4 What physical property is
Despite their structural differences, all c5h12 isomers have identical molecular formulas and. When the molar mass is indistinguishable for the isomers of ${c_5}{h_{12}}$. The physical property that is identical for the isomers of c5h12 is density. Isomers are compounds that have the same molecular formula but different structural formulas. Pentane, c5h 12, can form three structural isomers:
Solved What physical property is identical for the isomers
Isomers are compounds that have the same molecular formula but different structural formulas. When the molar mass is indistinguishable for the isomers of ${c_5}{h_{12}}$. Despite their structural differences, all c5h12 isomers have identical molecular formulas and. Density is a physical property that is defined as the mass of a. Butane, c4h 10, can form two.
Solved Density Questions and Problems Q4 What physical
Butane, c4h 10, can form two. Pentane, c5h 12, can form three structural isomers: Density is a physical property that is defined as the mass of a. When the molar mass is indistinguishable for the isomers of ${c_5}{h_{12}}$. Isomers are compounds that have the same molecular formula but different structural formulas.
Physical Properties Of Isomers Of C5H12
The physical property that is identical for the isomers of c5h12 is density. Butane, c4h 10, can form two. Pentane, c5h 12, can form three structural isomers: For the isomers of c5h12, which is. When the molar mass is indistinguishable for the isomers of ${c_5}{h_{12}}$.
Three Structural Isomers Have The Formula C5H12 Draw The, 58 OFF
Despite their structural differences, all c5h12 isomers have identical molecular formulas and. For the isomers of c5h12, which is. Density is a physical property that is defined as the mass of a. Isomers are compounds that have the same molecular formula but different structural formulas. The physical property that is identical for the isomers of c5h12 is density.
Physical Properties Of Isomers Of C5H12
For the isomers of c5h12, which is. Despite their structural differences, all c5h12 isomers have identical molecular formulas and. Density is a physical property that is defined as the mass of a. When the molar mass is indistinguishable for the isomers of ${c_5}{h_{12}}$. Butane, c4h 10, can form two.
When The Molar Mass Is Indistinguishable For The Isomers Of ${C_5}{H_{12}}$.
Pentane, c5h 12, can form three structural isomers: For the isomers of c5h12, which is. Isomers are compounds that have the same molecular formula but different structural formulas. Despite their structural differences, all c5h12 isomers have identical molecular formulas and.
The Physical Property That Is Identical For The Isomers Of C5H12 Is Density.
Density is a physical property that is defined as the mass of a. Butane, c4h 10, can form two.